What is a Lithium-ion Battery? How It Works Explained Simply
Lithium-ion batteries are the most widely used rechargeable (secondary) batteries in modern energy storage, powering everything from consumer electronics to electric vehicles and solar energy systems. But how do they actually work? Let’s break down their core chemical principles in simple terms.
What is a Lithium-ion Battery?
A lithium-ion battery is a type of rechargeable battery that relies on the movement of lithium ions (Li⁺) between its positive and negative electrodes to store and release energy. A key feature is that lithium always exists in the form of Li⁺ ions inside the battery, never as pure metallic lithium, which ensures safety and stability.
How Lithium-ion Batteries Work: Charging & Discharging
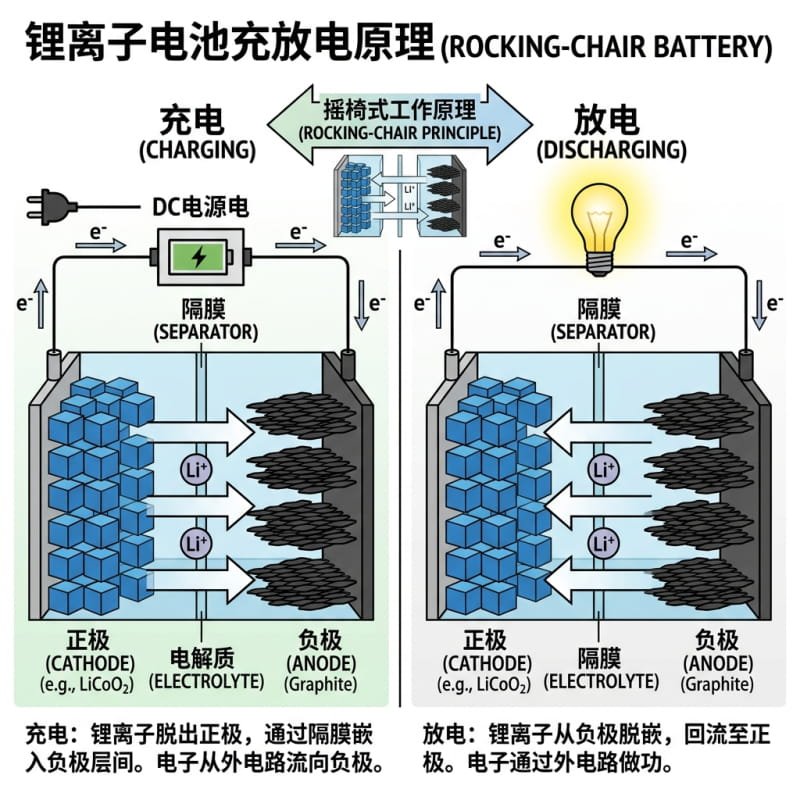
The working principle of lithium-ion batteries is often called the “rocking chair battery” because Li⁺ ions shuttle back and forth between the two electrodes during charge and discharge:
- – **Charging Process**: When you charge the battery, Li⁺ ions are extracted from the positive electrode, move through the electrolyte, and embed into the negative electrode (usually made of carbon/graphite). The negative electrode becomes lithium-rich, storing energy.
- – **Discharging Process**: When you use the battery, Li⁺ ions detach from the negative electrode, move back to the positive electrode through the electrolyte, and release electrical energy to power your devices. The more Li⁺ ions that return to the positive electrode, the higher the current and capacity of the battery.
Core Chemical Reactions
Taking lithium cobalt oxide (LiCoO₂) as the positive electrode material and graphite as the negative electrode material, the overall reversible redox reaction is:
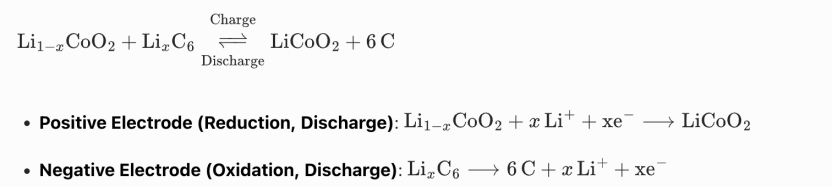
The charging process is the reverse of the above reactions. This excellent reversibility gives lithium-ion batteries outstanding cycle performance, high working voltage, and high specific energy.
Key Takeaways
- – Lithium-ion batteries are rechargeable, relying on Li⁺ ion movement between electrodes.
- – No metallic lithium is present in the battery, only Li⁺ ions.
- – The reversible redox reaction enables long cycle life and high energy density.