Lithium-Ion Battery Core Materials: A Complete Guide to Cathode, Anode, Electrolyte & Separator
Lithium-Ion Battery Core Materials: A Comprehensive Overview of the Four Pillars of Energy Storage
Introduction
As the core energy storage device of the new energy era, lithium-ion batteries have been deeply integrated into 3C consumer electronics, new energy vehicles (NEVs), energy storage stations, and other fields. The performance, lifespan, and safety of lithium-ion batteries are fundamentally determined by their four core raw materials: cathode materials, anode materials, electrolytes, and separators. This article will break down the “core skeleton” of lithium-ion batteries, helping you understand the function and selection logic of each material.
1. Cathode Materials: The “Heart” of Energy Density
Cathode materials serve as the “donor” of lithium ions in lithium-ion batteries, directly determining the upper limit of the battery’s energy density. They are typically micron-scale powder materials. Currently, the mainstream commercialized cathode materials mainly fall into four categories:
Lithium Cobalt Oxide (LiCoO₂): The first commercialized cathode material, featuring high voltage and high specific capacity. It is the traditional mainstream choice for 3C consumer electronics (mobile phones, laptops).
Lithium Manganese Oxide (LiMn₂O₄): Cost-effective, high in safety, and excellent in low-temperature performance, but with a shorter cycle life. It is widely used in low-end power batteries, energy storage batteries, and two-wheeler batteries.
Ternary Materials (LiNiₓMnᵧCo_zO₂): Combining the advantages of nickel, manganese, and cobalt, ternary materials can achieve high energy density or high safety by adjusting the element ratio. They are one of the core technical routes for NEV power batteries.
Lithium Iron Phosphate (LiFePO₄): Exceptionally high in safety, ultra-long in cycle life, and cost-competitive. It is the preferred material for energy storage stations and commercial vehicle power batteries, and also the fastest-growing cathode route in the current market.

2. Anode Materials: The “Storage Warehouse” for Lithium Ions
Anode materials act as the carrier for lithium ion intercalation/deintercalation during battery charging and discharging, directly affecting the battery’s charge-discharge efficiency and cycle life. The current mainstream anode material system includes:
Graphite Materials: Including natural graphite and artificial graphite, they are the most widely used anode materials today. With low lithium intercalation potential, excellent cycle stability, and low cost, graphite accounts for over 90% of the market share.
Hard Carbon/Soft Carbon Materials: Hard carbon offers excellent fast-charging performance and low-temperature performance, while soft carbon is suitable for high-rate discharge scenarios. Both are used in special batteries and high-end power batteries.
New-Generation Anode Materials: Lithium Titanate (high safety, long lifespan), Silicon-based Materials (ultra-high theoretical capacity), and Tin-based Materials are the core R&D directions for next-generation high-energy-density lithium-ion batteries.

3. Electrolyte: The “Blood” for Ion Transmission
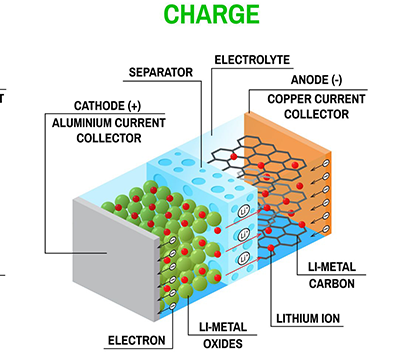
The electrolyte is the medium for lithium ion transmission between the cathode and anode inside the battery, equivalent to the “blood” of the battery. It directly affects the battery’s rate performance, low-temperature performance, and safety.
Core Composition: Consists of lithium salts, organic solvents, and additives. The commonly used lithium salt is Lithium Hexafluorophosphate (LiPF₆), and the organic solvent system is mostly a mixture of Ethylene Carbonate (EC), Dimethyl Carbonate (DMC), Diethyl Carbonate (DEC), and Ethyl Methyl Carbonate (EMC). The ratio is adjusted to meet different battery requirements.
Classification: Liquid electrolytes are mainstream, while new systems such as gel electrolytes and solid electrolytes are developing rapidly.
4. Separators & Auxiliary Materials: The “Barrier” for Battery Safety
Separator: Located between the cathode and anode, it plays a key role in isolating electrons and conducting lithium ions, serving as the core line of defense for battery safety. The mainstream separators are polyethylene (PE) and polypropylene (PP) single-layer/multi-layer porous membranes, which possess excellent chemical stability and thermal stability.
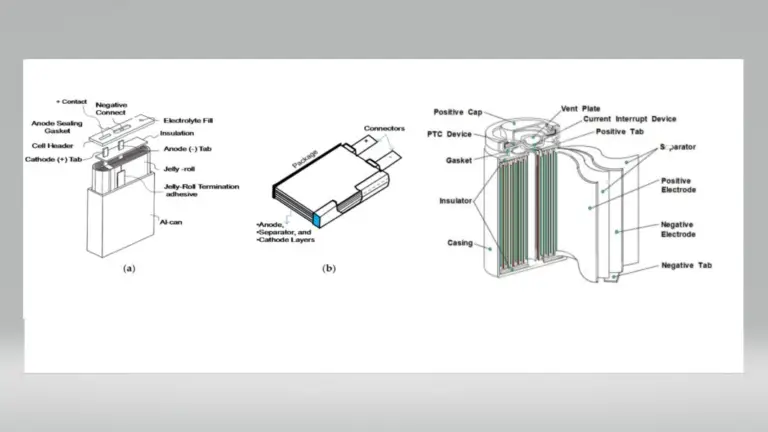
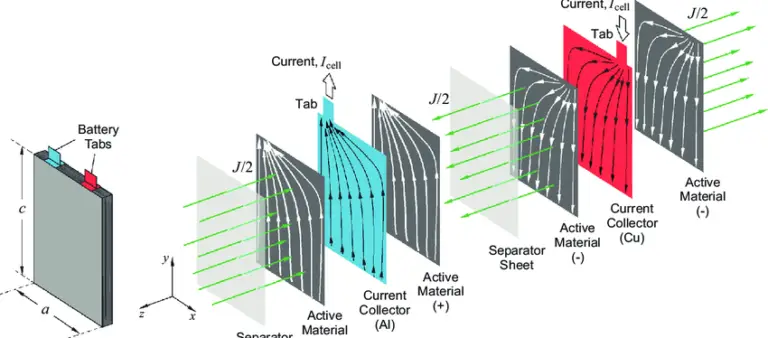
Auxiliary Materials: Including conductive agents (carbon black, VGCF, carbon nanotubes), binders (PVDF, SBR), current collectors (aluminum foil for cathodes, copper foil for anodes), and battery casings (aluminum-plastic composite film, aluminum casings, steel casings). These materials are crucial for ensuring the structural stability and conductive performance of the battery.
Conclusion
Each of the four core materials of lithium-ion batteries directly determines the upper limit of battery performance. From lithium cobalt oxide in the 3C field to ternary and lithium iron phosphate in power batteries, and the lithium iron phosphate route in energy storage, material selection is driven by both technical and market factors. In the next article, we will deeply analyze the technical route competition of core materials and explore the future trends of the industry.