Analysis of Factors Affecting Internal Resistance of Lithium-Ion Batteries
Lithium-ion battery internal resistance is one of the most important indicators affecting battery efficiency, charging performance, thermal stability, and cycle life. In electric vehicles, renewable energy storage systems, industrial power equipment, and portable electronics, internal resistance directly influences power output capability and overall operational reliability.
As Lithium-ion battery technology continues advancing, understanding the factors that affect battery internal resistance has become essential for battery manufacturers, engineers, and energy system designers.
Explore LNC Batteries Company specialize in advanced battery solutions, with expertise in Lithium-ion technologies, including LiFePO4, Li-ion, Li-polymer, as well as Lithium batteries like LiMnO2 and LiSOCl2, and Na-ion batteries. Quality guaranteed.
What Is Lithium-Ion Battery Internal Resistance
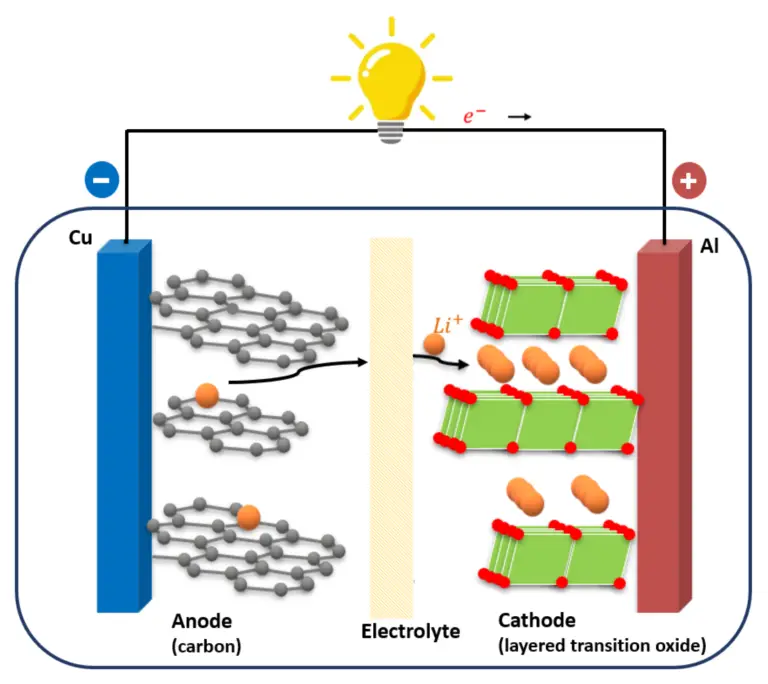
Internal resistance refers to the resistance encountered by current flow inside a battery during charging and discharging processes. It consists of electronic resistance, ionic resistance, and electrochemical reaction resistance.
When internal resistance increases, battery efficiency decreases, heat generation rises, and voltage drop becomes more significant under load conditions.
The voltage drop relationship can be expressed as:
Voltage Drop (V)=Current (A)×Internal Resistance (Ω)
A lower internal resistance generally improves battery power delivery and charging efficiency.
Types of Internal Resistance
Lithium-ion battery internal resistance can be divided into several categories.
Ohmic Resistance
Ohmic resistance originates from electrode materials, current collectors, electrolyte conductivity, and connection interfaces.
This resistance is relatively stable and mainly depends on material conductivity.
Charge Transfer Resistance
Charge transfer resistance occurs during electrochemical reactions between electrodes and electrolyte interfaces.
This resistance changes dynamically during battery operation and strongly affects charging and discharging efficiency.
Diffusion Resistance
Diffusion resistance is related to lithium-ion movement inside active materials.
As ion diffusion slows down, internal resistance increases, especially during high-rate charging or low-temperature operation.
Temperature Effects on Battery Internal Resistance
Temperature is one of the most critical factors affecting Lithium-ion battery internal resistance.
Low Temperature Increases Resistance
At low temperatures, electrolyte viscosity increases and lithium-ion mobility decreases.
This slows electrochemical reactions and raises internal resistance significantly.
The battery power equation remains:
Power (W)=Voltage (V)×Current (A)
When internal resistance rises, voltage loss increases, reducing effective battery power output.
High Temperature Accelerates Degradation
Although moderate temperature increases may temporarily reduce resistance, excessive heat accelerates electrolyte decomposition and electrode aging.
Long-term exposure to high temperatures can permanently increase battery internal resistance and shorten cycle life.
Explore LNC Batteries Company specialize in advanced battery solutions, with expertise in Lithium-ion technologies, including LiFePO4, Li-ion, Li-polymer, as well as Lithium batteries like LiMnO2 and LiSOCl2, and Na-ion batteries. Quality guaranteed.
Influence of Electrode Materials
Battery electrode materials directly affect conductivity, electrochemical reaction speed, and ion transport efficiency.
Cathode Material Structure
Different cathode materials provide different conductivity characteristics and internal resistance performance.
Materials with higher conductivity and stable crystal structures generally produce lower internal resistance during cycling.
LiFePO4 batteries are known for strong thermal stability, while some high-energy Lithium-ion chemistries focus more on maximizing energy density.
Anode Material Characteristics
Graphite remains the most common anode material, but silicon-enhanced anodes are increasingly used to improve capacity performance.
However, repeated expansion and contraction of silicon materials may affect long-term resistance stability.
Electrode Coating Uniformity
Uneven electrode coating thickness may create inconsistent current distribution inside battery cells.
This can increase localized resistance and accelerate internal heat generation during operation.
Electrolyte and Separator Impact
Electrolytes and separators play important roles in maintaining efficient ion transport inside Lithium-ion batteries.
Electrolyte Conductivity
Electrolytes with higher ionic conductivity help reduce internal resistance and improve charging efficiency.
Electrolyte composition also affects battery performance under high-temperature and low-temperature conditions.
Separator Porosity
Battery separators control ion flow between electrodes while preventing internal short circuits.
Improved separator porosity enhances ion mobility and reduces diffusion resistance.
Effect of Charge and Discharge Conditions
Operating conditions strongly influence short-term and long-term internal resistance behavior.
High C-Rate Charging and Discharging
High current operation increases polarization effects and internal heat generation.
The charging current formula is:
Charging Current (A)=Battery Capacity (Ah)×C-Rate
As C-rate increases, battery internal resistance becomes more apparent due to faster ion movement and increased electrochemical stress.
Depth of Discharge
Deep discharge conditions place greater stress on electrode materials and may accelerate internal structural degradation.
Repeated deep cycling often contributes to gradual resistance growth over time.
Battery Aging and Internal Resistance Growth
As Lithium-ion batteries age, internal resistance naturally increases due to material degradation and chemical side reactions.
SEI Layer Growth
The solid electrolyte interphase layer forms on the anode surface during battery operation.
While this layer helps stabilize the battery, excessive SEI growth increases ion transport resistance.
Electrode Structural Damage
Repeated charging and discharging cycles create microscopic cracks and mechanical stress inside electrode materials.
This structural degradation gradually reduces conductivity and increases resistance.
Electrolyte Decomposition
Electrolyte aging generates byproducts that interfere with ion transport and reduce electrochemical efficiency.
Over time, this contributes to higher heat generation and reduced battery performance.
Explore LNC Batteries Company specialize in advanced battery solutions, with expertise in Lithium-ion technologies, including LiFePO4, Li-ion, Li-polymer, as well as Lithium batteries like LiMnO2 and LiSOCl2, and Na-ion batteries. Quality guaranteed.
Methods for Reducing Lithium-Ion Battery Internal Resistance
Battery manufacturers continue developing technologies to minimize internal resistance and improve efficiency.
Advanced Conductive Materials
Improved conductive additives and optimized electrode structures help reduce electronic resistance inside battery cells.
Optimized Electrolyte Formulations
New electrolyte technologies improve ion conductivity and thermal stability under demanding operating conditions.
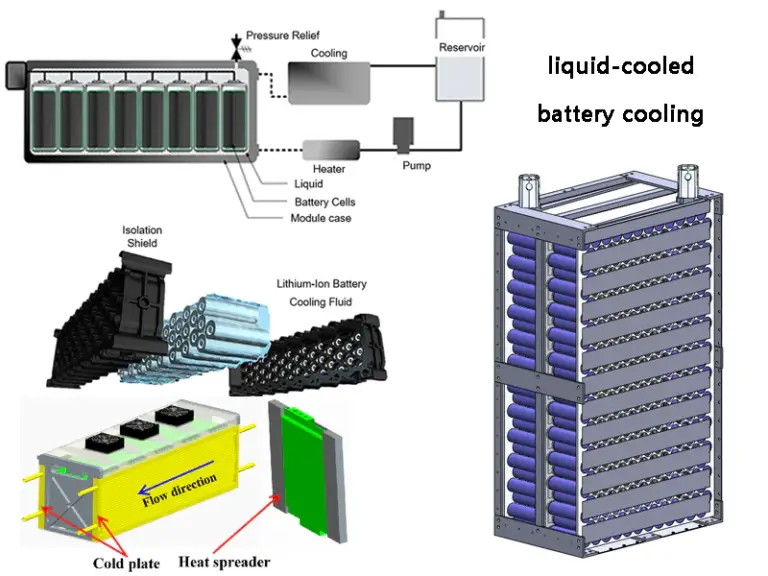
Improved Thermal Management Systems
Effective cooling systems help maintain stable battery temperature and reduce resistance fluctuations during high-power operation.
As Lithium-ion technology evolves, reducing internal resistance remains one of the key objectives for improving battery efficiency, charging speed, and long-term operational stability.